Epidemiology
- The 6th most common malignancy in women worldwide.
- The 4th most common malignancy in women in developed countries.
- The most common gynecological malignancy in the western world.
- The median age of diagnosis is 63 years, while >90% of the cases involve women above 50 years of age, when 4% of cases involve patients below 40 years.
- 90-95% sporadic, 5-10% hereditary,
- Hereditary cases: mostly autosomal-dominant inherited cancer susceptibility syndrome called HNPCC syndrome or Lynch syndrome type II (DNA Mismatch-Repair MMR gene mutations), or the rare Cowden’s syndrome (phosphatase and tensin homolog PTEN mutation).
- 90% are endometrial adenocarcinomas, the rest are papillary serous, clear cell, papillary endometrial, mucinous carcinomas.
- The uterine carcinosarcoma is a high grade endometrial cancer variant (“like grade 4 out of 3”) that represents about 5% of all uterine corpus cancers and accounts for 15% of the mortality.
Factors that Increase the Risk
- Oral intake of estrogens (without progestins),
- Early menarche,
- Late menopause,
- Low parity,
- Extended periods of anovulation
- Obesity
- Increasing age
- Genetic predisposition (HNPCC, Cowden)
- PCOS
- SERMs tamoxifen and raloxifene
- Sedentary lifestyle
- Diabetes mellitus
- Hypertension
Factors that Decrease the Risk
- Combined oral contraceptives
- Parity
- Late age at last birth
- Physical activity
- Metformin use
Staging
FIGO 2009
- IA : Zero or <50% myometrial invasion
- IB: Invasion ≥ 50% of the myometrium
- II: Tumor invades cervical stroma, but does not extend beyond the uterus
- IIIA: Tumor invades the serosa of the corpus uteri and/or adnexa
- IIIB: Vaginal and/or parametrial involvement
- IIIC1: Positive pelvic lymph node
- IIIC2: Positive paraaortic lymph node
- IVA: Invasion of bladder and/or bowel mucosa
- IVB: Distant metastasis
Diagnosis
Clinical Image
90% of cases present with abnormal uterine bleeding. Sometimes incidental finding of enlarged uterus (hematometra). Rarely presentation with hydronephrosis, hematuria or melena in a late stage.
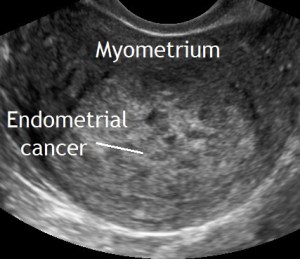
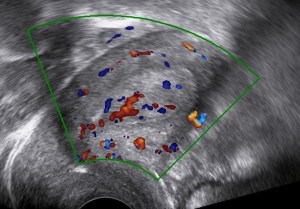
Ultrasound
The following ultrasound features may be associated with endometrial cancer:
- Thick endometrium (>3mm in the postmenopause)
- Large tumor volume
- Irregular endometrial-myometrial junction
- Non-uniform echogenicity
- Multiple vessels
Always bear in mind that endometrial cancer can also be found within adenomyosis.
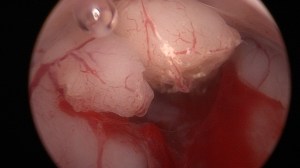
Hysteroscopy with biopsy
Remains the gold standard in providing the histopathological diagnosis.
Screening
There is currently no evidence to support the screening of asymptomatic women. However one should not underestimate the value of ultrasound (i.e. patients with cervical stenosis and hematometra).
Patients with Lynch syndrome Type II besides having regular colonoscopies should at least keep a menstrual calendar and report abnormal bleeding. Furthermore annual screening with an endometrial biopsy starting at age 35 is justifiable. A reasonable case for the prophylactic hysterectomy with salpingectomy can be made as early as at 40 years of age, as the lifetime endometrial cancer risk in women with Lynch syndrome type II is about of 30-60%.
Patients taking tamoxifen should have an endometrial biopsy if they present with abnormal bleeding.
Types
- Type 1: Estrogen-dependent endometrioid adenocarcinomas 80% of cases.
- Type 2: Estrogen-independent non-endometrioid carcinomas (serous, clear-cell, undifferentiated).
Treatment
- Surgery
- Minimal-invasive surgery is preferred in early stage disease.
- Standard surgery is total hysterectomy with bilateral salpingooophorectomy without vaginal cuff resection.
- Staging infracolic omentectomy should be performed in clinical stage I serous endometrial carcinoma, carcinosarcoma, and undifferentiated carcinoma. It can be omitted in clear cell and endometrioid carcinoma in stage I disease.
- Lymphadenectomy
- No myometrial invasion: no need for lymphadenectomy.
- Low- and intermediate- risk disease: sentinel lymphadenectomy can be considered / systematic lymphadenectomy is not recommended.
- High-intermediate- and high-risk disease: systematic lymphadenectomy should be performed / sentinel lymphadenectomy is an accepted alternative for stage I and II disease.
- Fertility-Sparing: may only be considered for endometrioid endometrial cancer G1 without myometrial involvement which has been hysteroscopically excised and the patient is adjuvantly treated with progestins and strictly followed-up.
- Radiation
- Brachytherapy
- Can be recommended to reduce vaginal cuff recurrence from intermediate-risk disease onward.
- EBRT (External Beam Radiation Therapy)
- From high-intermediate-risk disease onward.
- Brachytherapy
- Chemotherapy
- From high-intermediate-risk disease onward, typically with Carboplatin and Paclitaxel.
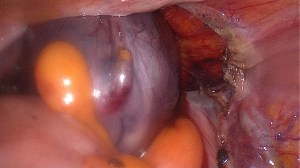
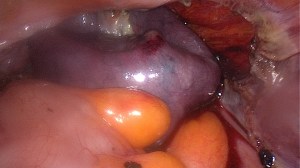
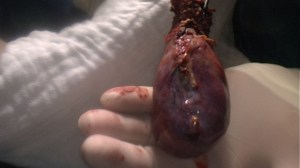
The above images show a case of a giant hematometra in a case of cervical stenosis and mucinous endometrial cancer. See how the uterus shrinks after its contents have been safely evacuated. There was no difficulty removing it afterwards.
Prognostic Factors
- Stage
- Grade
- LVSI Lymph-vascular space invasion
- Histological subtype
Follow-Up
Gynecological examination and ultrasound
- Years 1-3: every 3 months
- Years 4-5: every 6 months
Bibliography
- Colombo, N., Preti, E., Landoni, F., Carinelli, S., Colombo, A., Marini, C., Sessa, C., & ESMO Guidelines Working Group (2013). Endometrial cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of oncology : official journal of the European Society for Medical Oncology, 24 Suppl 6, vi33–vi38. https://doi.org/10.1093/annonc/mdt353
- Raglan, O., Kalliala, I., Markozannes, G., Cividini, S., Gunter, M.J., Nautiyal, J., Gabra, H., Paraskevaidis, E., Martin‐Hirsch, P., Tsilidis, K.K. and Kyrgiou, M. (2019), Risk factors for endometrial cancer: An umbrella review of the literature. Int. J. Cancer, 145: 1719-1730. https://doi.org/10.1002/ijc.31961
- Cantrell, L. A., Blank, S. V., & Duska, L. R. (2015). Uterine carcinosarcoma: A review of the literature. Gynecologic oncology, 137(3), 581–588. https://doi.org/10.1016/j.ygyno.2015.03.041
- Biller, L. H., Syngal, S., & Yurgelun, M. B. (2019). Recent advances in Lynch syndrome. Familial cancer, 18(2), 211–219. https://doi.org/10.1007/s10689-018-00117-1
- Epstein, E. , Fischerova, D. , Valentin, L. , Testa, A. C., Franchi, D. , Sladkevicius, P. , Frühauf, F. , Lindqvist, P. G., Mascilini, F. , Fruscio, R. , Haak, L. A., Opolskiene, G. , Pascual, M. A., Alcazar, J. L., Chiappa, V. , Guerriero, S. , Carlson, J. W., Van Holsbeke, C. , Giuseppe Leone, F. P., De Moor, B. , Bourne, T. , van Calster, B. , Installe, A. , Timmerman, D. , Verbakel, J. Y. and Van den Bosch, T. (2018), Ultrasound characteristics of endometrial cancer as defined by International Endometrial Tumor Analysis (IETA) consensus nomenclature: prospective multicenter study. Ultrasound Obstet Gynecol, 51: 818-828. doi:10.1002/uog.18909
- Breijer, M. C., Peeters, J. A., Opmeer, B. C., Clark, T. J., Verheijen, R. H., Mol, B. W. and Timmermans, A. (2012), Capacity of endometrial thickness measurement to diagnose endometrial carcinoma in asymptomatic postmenopausal women: a systematic review and meta‐analysis. Ultrasound Obstet Gynecol, 40: 621-629. doi:10.1002/uog.12306
- Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF): Diagnostik, Therapie und Nachsorge der Patientinnen mit Endometriumkarzinom, Langversion 1.0, 2018, AWMF Registernummer: 032/034-OL, http://www.leitlinienprogramm-onkologie.de/leitlinien/endometriumkarzinom/
(abgerufen am: 02.07.2018). - Naftalin, J. , Nunes, N. , Hoo, W. , Arora, R. and Jurkovic, D. (2012), Endometrial cancer and ultrasound: why measuring endometrial thickness is sometimes not enough. Ultrasound Obstet Gynecol, 39: 106-109. doi:10.1002/uog.9062.
- Concin, N., Matias-Guiu, X., Vergote, I., Cibula, D., Mirza, M. R., Marnitz, S., Ledermann, J., Bosse, T., Chargari, C., Fagotti, A., Fotopoulou, C., Gonzalez Martin, A., Lax, S., Lorusso, D., Marth, C., Morice, P., Nout, R. A., O’Donnell, D., Querleu, D., Raspollini, M. R., … Creutzberg, C. L. (2021). ESGO/ESTRO/ESP guidelines for the management of patients with endometrial carcinoma. International journal of gynecological cancer : official journal of the International Gynecological Cancer Society, 31(1), 12–39. https://doi.org/10.1136/ijgc-2020-002230